Vertex/CRISPR to begin rolling review in US for gene-edited therapy; get FDA designations | Seeking Alpha

Y-mAbs Reports Initiation of Rolling Review of BLA for Naxitamab to the US FDA to Treat Neuroblastoma
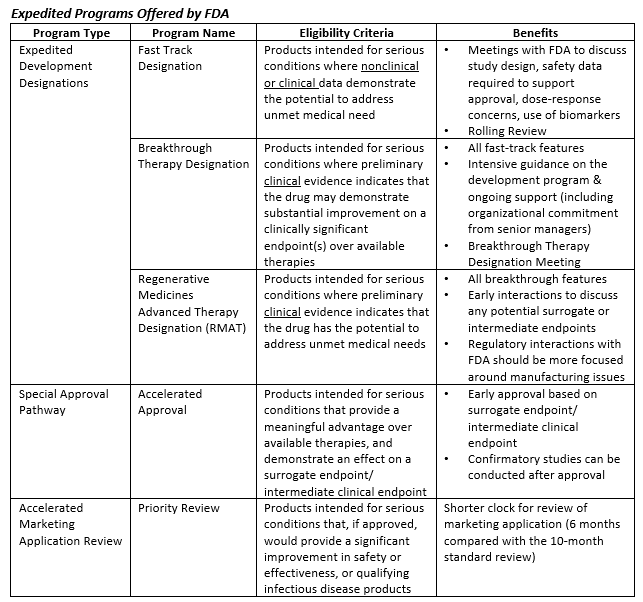
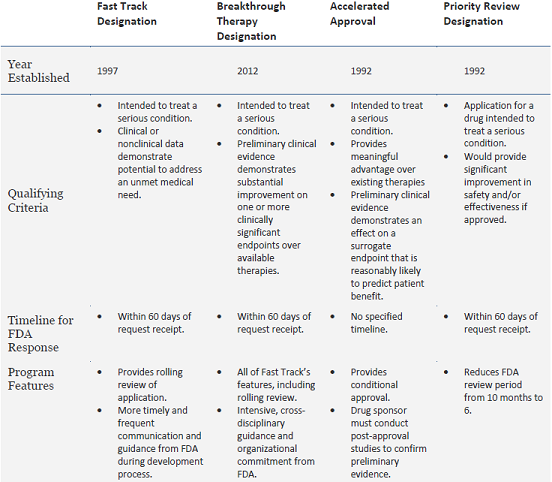
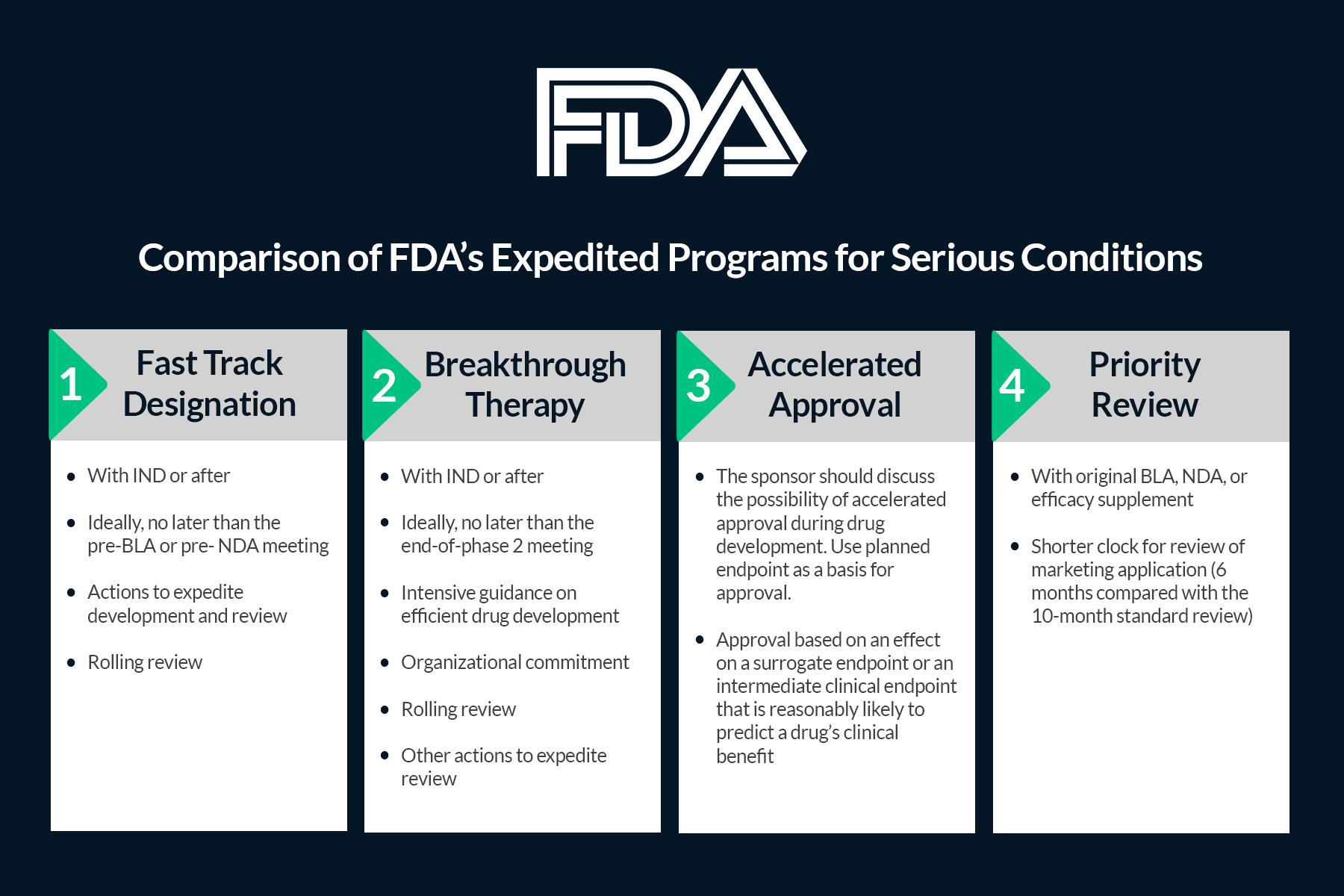
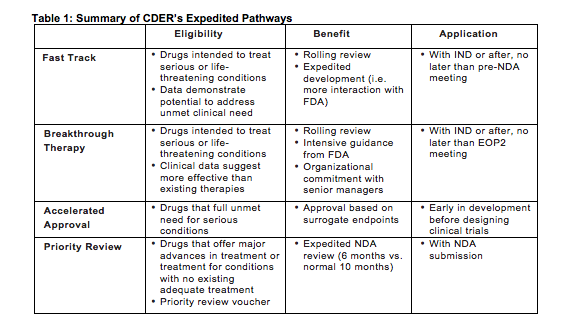
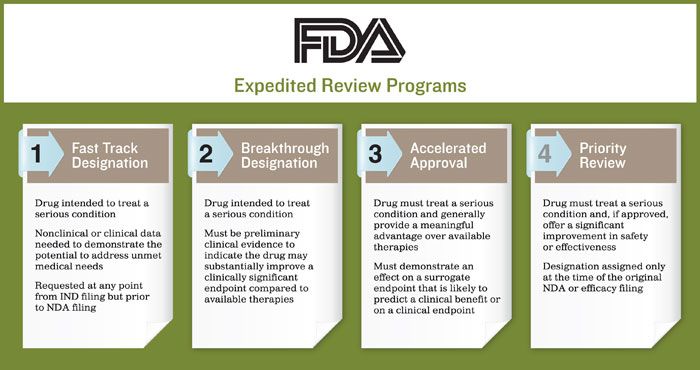
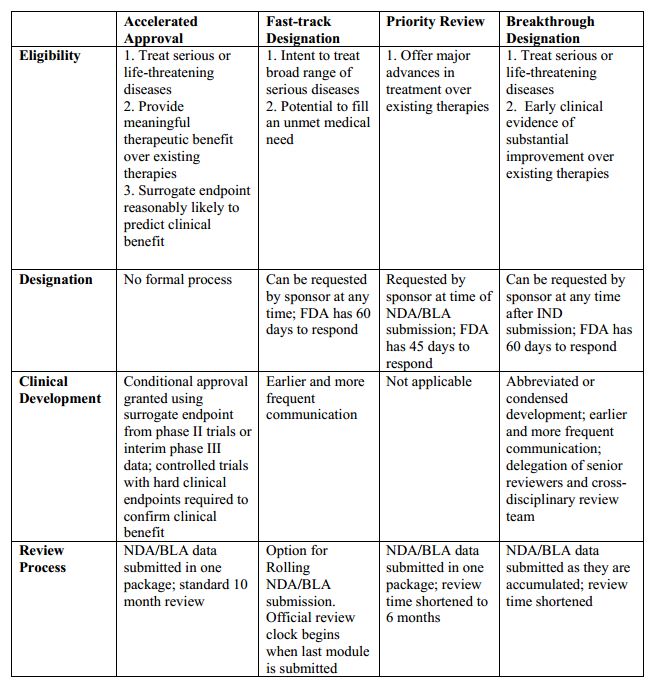
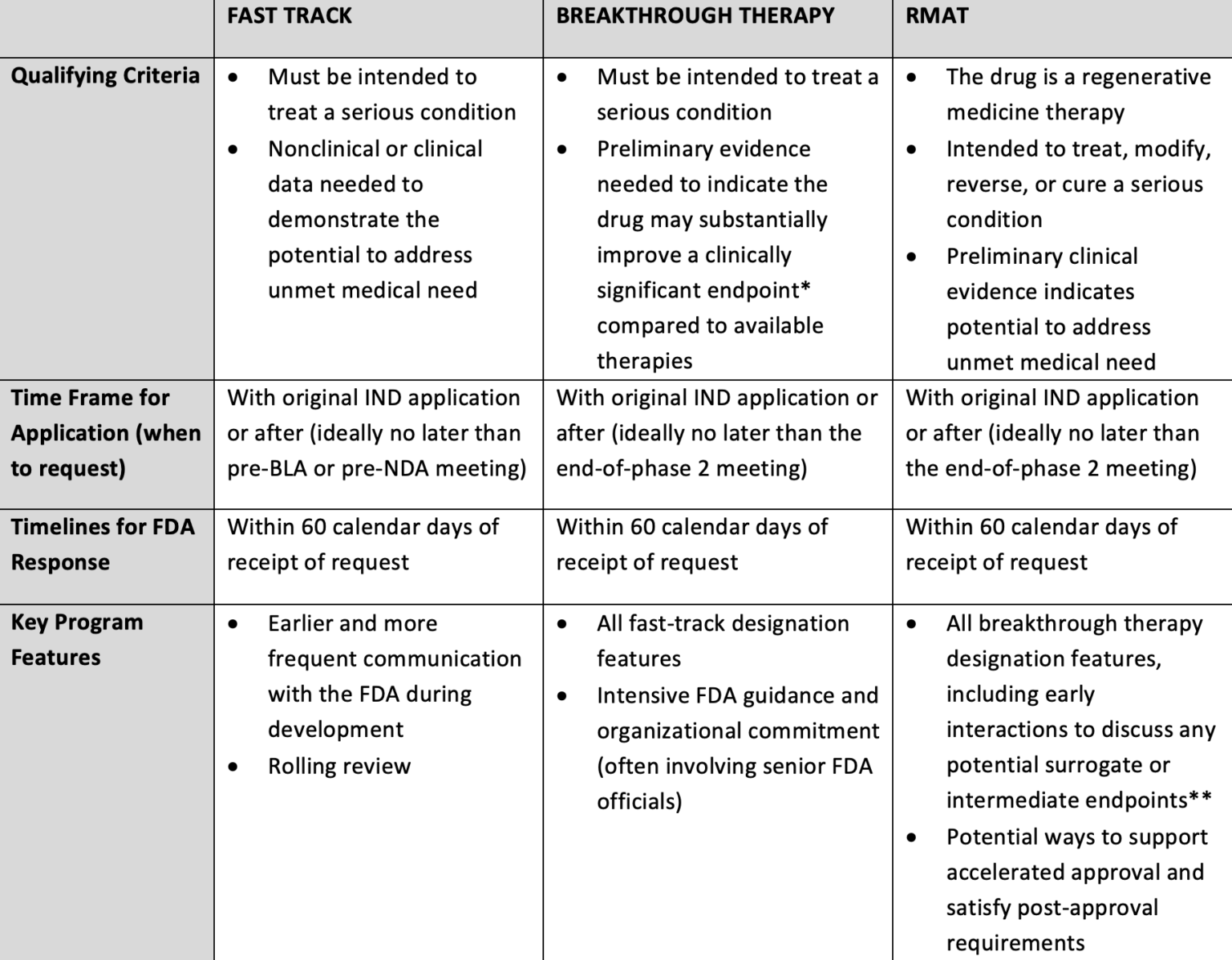
The Need for Speed in Drug Development: A Sponsor's Guide to FDA Expedited Programs | Halloran Consulting Group
FDA Moves Against Fast Track Vaccines - News about Energy Storage, Batteries, Climate Change and the Environment

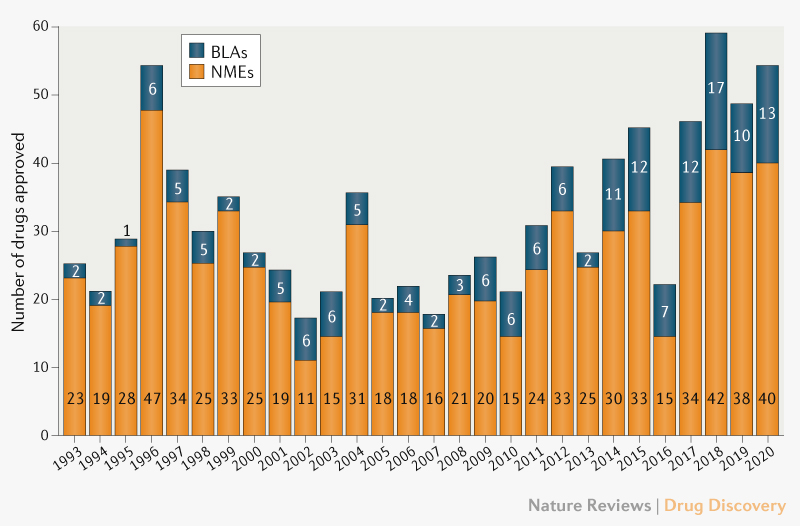
Analysis of the Real-Time Oncology Review (RTOR) Pilot Program for Approvals of New Molecular Entities | SpringerLink

CytoDyn Inc. submits the first of three main sections of its HIV Biologics License Application to FDA under rolling review
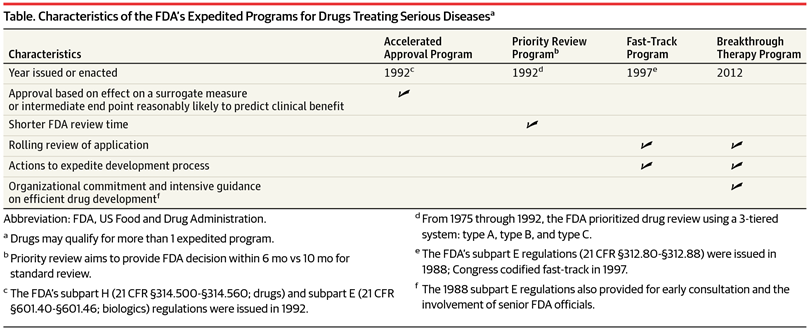
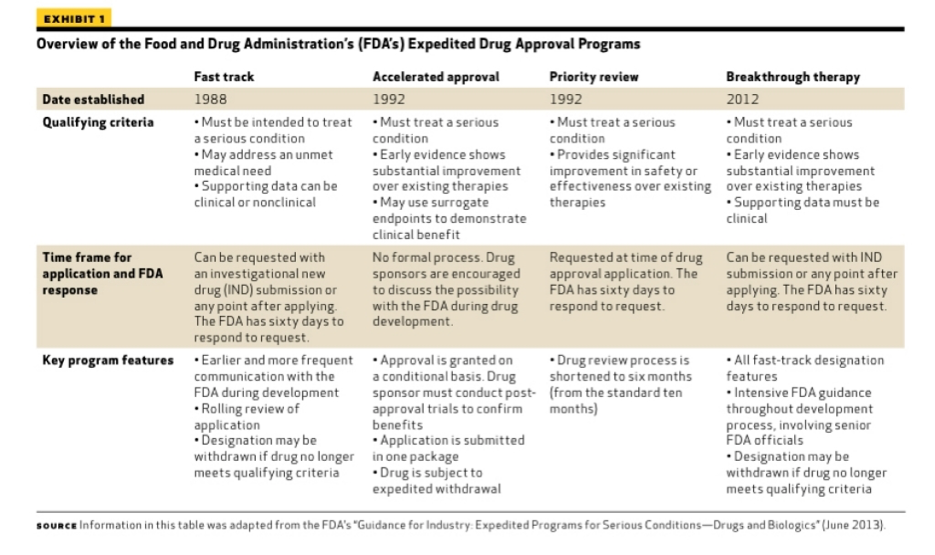
The Science Of A Biotech Valuation: How To Interpret The Value Of FDA Expedited Programs (NASDAQ:IBB) | Seeking Alpha

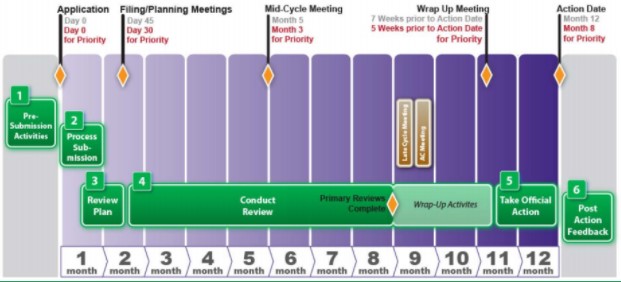
/cloudfront-us-east-2.images.arcpublishing.com/reuters/LUSOBJZ5H5OIDD7VBNDDQP62F4.jpg)